Those 65 and older were also slightly more likely to have grade 3 or higher side effects. This side effect shows up in lab readings and could cause bleeding problems such as nosebleeds, but overall it was unlikely to be noticed by participants.Īsians were more likely to have grade 3 or higher side effects, especially low blood platelet count. The most common grade 3, severe, or higher, side effect was low blood platelet count, seen in 11.9 percent of participants. Low blood platelet count (32.2 percent).The most common side effects of any severity level were: This study found 17.2 percent of people needed their dose changed because of side effects and 7 percent stopped treatment because of side effects. Those studies featured 884 people who took at least one dose of T-DM1. Researchers looked at side effect information from seven studies involving T-DM1. These researchers used that data to look at the safety of T-DM1 on a large scale. The EMILIA study and other research involving T-DM1 has created a large pool of information about how people react to the medicine. T-DM1 was approved by the FDA for treatment of HER2-positive, metastatic breast cancer in 2013 after the EMILIA study showed it did a better job of lengthening life and stopping the cancer from growing than standard treatments.
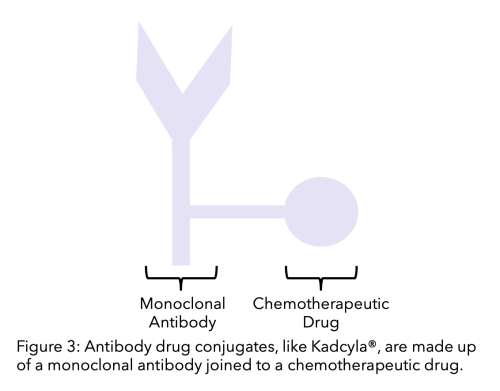

This medicine is used to treat HER2-positive breast cancer, which grows when breast cells have too many HER2 proteins. In this case, the targeted therapy is trastuzumab (Herceptin). This causes fewer side effects than when the two medicines are given separately.

T-DM1 is an antibody-drug conjugate, a treatment that pairs a chemotherapy medicine with a targeted therapy to get the chemotherapy directly to tumor cells. If they did occur, they were usually manageable. After looking at information from seven studies involving T-DM1 (Kadcyla), researchers found that severe side effects were uncommon.
